Lieben反應是指在鹵素和氫氧化鈉(次氯酸鈉)作用下,把甲基酮轉化為羧酸鹽和鹵仿的反應,也稱鹵仿反應。如果所用的鹵素為碘,則稱為碘仿反應,常用作鑒別甲基酮的顯色反應。
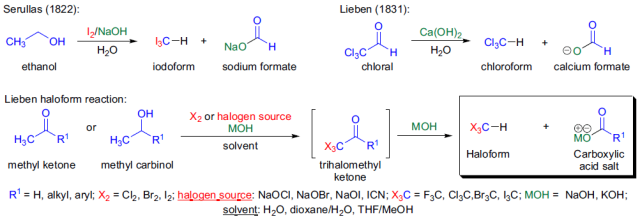
反應機理
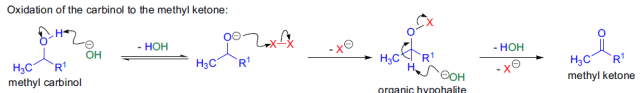
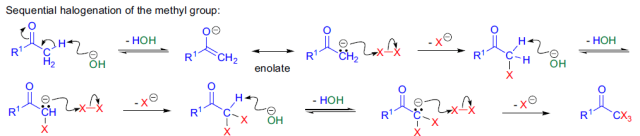
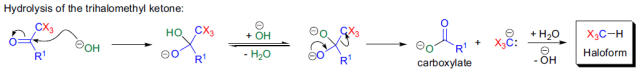
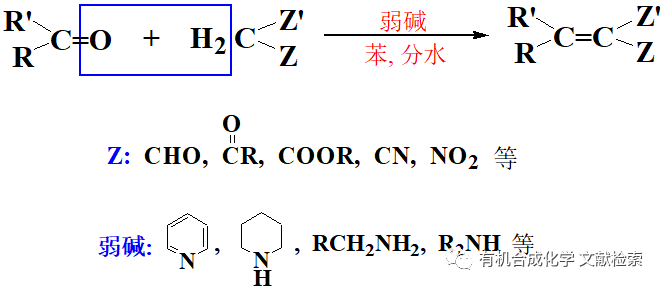
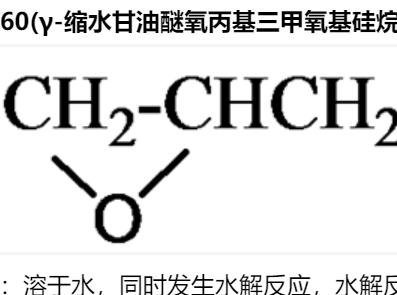
由上述反應機理可以看出,反應中氫氧化鈉除了作為堿外,還作為親核試劑。如果使用其它堿,使反應在無水條件下進行,則反應生成的鹵仿碳負離子可作為親核試劑與其它親電試劑反應,這是Langlois建立的三氟甲基化方法。
反應實例
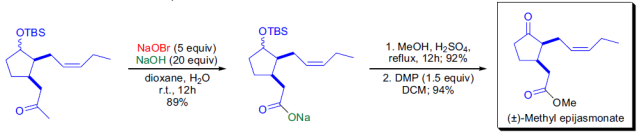
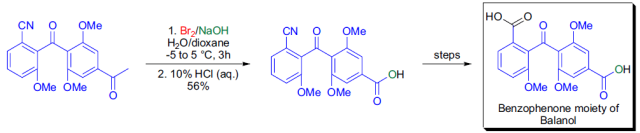
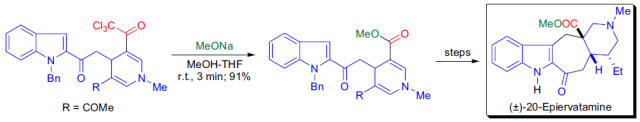
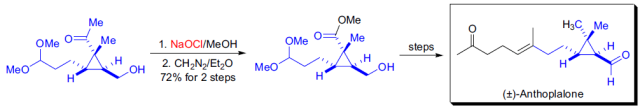
參考文獻
1. Serullas. Effect of iodine on thebasic solution of alcohols. Ann. chim. (Paris) [13] 1822, 20, 165.
2. von Liebig, J. Reaction ofalcohols with chlorine gas. Ann. Phys. Chem. Ser. 2 1831, 23, 444.
3. Lieben, A. The formation ofiodoform and the application of this reaction in the chemical analysis. Liebigs Ann.Chem. 1870, Supp. 7, 218-236.
4. Hailes, H. C., Isaac, B., HashimJavaid, M. Synthesis of methyl epijasmonate and cis-3-(2-oxopropyl)-2-(pent-2Z-enyl)-cyclopentan-1-one.Tetrahedron 2001,57, 10329-10333.
5. Storm, J. P., Andersson, C.-M.Iron-Mediated Synthetic Routes to Unsymmetrically Substituted, StericallyCongested Benzophenones. J. Org. Chem. 2000, 65, 5264-5274.
6. Bennasar, M. L., Vidal, B.,Bosch, J. Total Synthesis of Indole Alkaloids of the Ervatamine Group. ABiomimetic Approach. J. Org. Chem.
1996,61, 1916-1917.
7.Ihara, M., Taniguchi, T., Tokunaga, Y., Fukumoto, K. Ring Contraction ofCyclobutanes and a Novel Cascade Reaction: Application to Synthesis of (±)-Anthoplalone and (±)-Lepidozene. J. Org. Chem. 1994, 59, 8092-8100.
摘自:有機人名反應——機理及應用 第四版,Strategic Applications of Named Reactions in Organic Synthesis