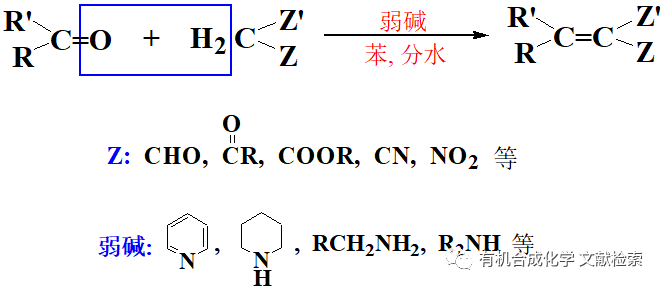
1927 年,同樣是德國的化學家G.Balz和G.Schiemann發現直接 加熱苯胺的硼氟酸重氮鹽能得到氟苯,這就是Balz-Schiemann反應。1935 年, F.B.Dains 和 F.Eberly用KI去處理重氮鹽,成功合成了碘代苯。隨后重氮化羥基取代和重氮化去胺反應也相繼被發現,加上偶氮反應,形成了比較完善的芳香重氮化合物反應體系。
在芳香重氮鹽的碘代反應中,可不必加入銅鹽作催化劑,只需和KI或NaI直 接反應。因為I- 的氧化還原電位(E0 = 1.3 V)足以促使電子向重氮離子轉移,而使反應順利進行。這個反應實際上Griess早在 1864 年就發現了。反應產率一般比氯代和溴代都高,反應也容易,重氮化完成后直接加入KI在室溫下就能發生反應。
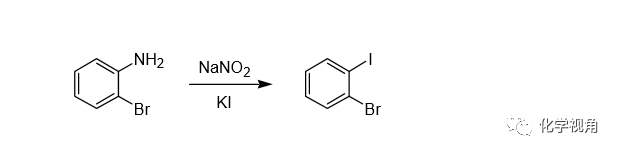
In a 600-ml. beaker fitted with a thermometer and mechanical stirrer are placed 150 ml. of concentrated hydrochloric acid and 55 g. (0.32 mole) of o-bromoaniline. After brief stirring, 100 g. of ice is added and the beaker is surrounded by an ice-salt bath. The solution is then diazotized by the dropwise addition with stirring of a solution of 24.3 g. (0.35 mole) of sodium nitrite in 100 ml. of water, the temperature being kept at 0–5°c.
After stirring the diazotized solution for 15 minutes, it is slowly poured through a glass-wool filter into a solution of 180 g. (3.4 moles) of potassium iodide in 600 ml. of water. After standing overnight, the heavy dark oil is separated, washed successively with 10% aqueous sodium hydroxide, water, 5% aqueous sodium bisulfite and water, and then dried over magnesium sulfate.
Distillation under reduced pressure gives o-bromoiodobenzene as a nearly colorless liquid, b.p. 120–121° at 15 mm. Yield 65–75 g. (72–83%).