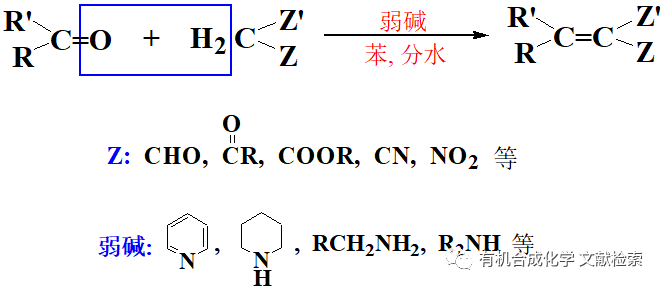
▲共同第一作者:楊絲雨,丘榮星
通訊作者:龐元杰教授
通訊單位:華中科技大學
論文DOI:10.1021/acscatal.5c03184
我們采用加壓CO來提高近表面CO濃度,從而引發催化劑動態重構,生成更多 Cu(111)/Cu(100)界面以增強C1-C2耦合。當在3 atm CO壓力下進行CO還原反應并伴隨原位催化劑重構時,Cu(111)/Cu(100)催化活性界面的活性比常壓條件下高出1.7倍;在不同CO壓力下,Cu(111)/Cu(100)界面密度與正丙醇選擇性之間建立了定量相關性。

HF-Cu預催化劑在不同CO壓強(1、3、10 atm)下進行重構的催化劑,在還原重建過程中,增加CO壓力對形貌的影響很小,但Cu晶面尺寸和晶面類型的比例發生了變化。

12px;">(a-c) SEM of the HF-Cu-1, HF-Cu-3, and HF-Cu-10 catalysts after CORR, respectively. (d-f) TEM of the HF-Cu-1, HF-Cu-3, and HF-Cu-10, respectively. (g) Cu LMM of the HF-Cu-x catalyst. (h) XRD of the HF-Cu-x catalyst. (i) OHads of the HF-Cu-x catalyst in 1 M KOH.
進一步研究重構后的Cu晶面的空間分布,與HF-Cu-1相比,HF-Cu-3和HF-Cu-10具有更多個碎片和更小的晶面。在不同壓力下量化單個Cu(111)和Cu(100)面的面積,HF-Cu-3和HF-Cu-10的Cu(111)小平面面積顯著減少。這一觀察結果表明,在高CO濃度下,Cu晶面會進行動態重構,從而產生更小、更碎片化的結構。我們進一步量化了不同壓力下單位面積Cu(111)/Cu(100)界面的界面密度,HF-Cu-1、HF-Cu-3和HF-Cu-10的界面密度分別為26 μm–1,45 μm–1和36 μm–1,這意味著更多的Cu(111)/Cu(100)界面可以促進*C1和*C2中間體耦合。

(a-c) HRTEM images show facet information for the HF-Cu-1, HF-Cu-3, and HF-Cu-10, respectively. As visual aids, yellow and red dotted lines circle fragments of the Cu(100) and Cu(111) facets and green dotted lines highlight the interfaces between the (100) and (111) facets. FFTs of two typical areas are shown one for the (100) facet (A) and the other for the (111) facet (B). (d-f) The Cu(100) and Cu(111) facets on each sample are highlighted of the HF-Cu-1, HF-Cu-3, and HF-Cu-10, respectively. (g) Two typical profiles of integrated pixel intensity profiles labeled for Cu(111) (0.210 nm) and Cu (100) (0.180 nm). (h) Each sample from 6 individual HRTEM micrographs, covering the area of individual Cu(111) and Cu(100) facets from the HF-Cu-x. (i) Cu(111)/Cu(100) interfaces per unit area of HF-Cu-x.
隨后研究了HF-Cu-x催化劑在不同CO壓力下的CORR性能,當在-0.67 V的恒定電位下進行CORR實驗時,在1至10 atm的壓力范圍內,正丙醇FE呈現火山形趨勢,HF-Cu-3產正丙醇的FE為28%時達到峰值(即HF-Cu系列材料在3 atm的CO壓力下表現出最高的正丙醇FE)。此外,FE正丙醇/FEC2+比率呈火山狀趨勢,從25%上升到39%,然后下降,HF-Cu-4實現最大FE正丙醇/FEC2+比率。這些發現表明,升高的CO壓力通過加速*C1和 *C2的動力學偶聯實現對正丙醇合成的偶聯。此外,在MEA中進行了穩定性測試,-100mA cm-2的電流密度下,正丙醇的FE在最初的50 h內保持增加趨勢,之后變得穩定,在80 h的持續時間內平均達到20%以上。HF-Cu-3催化劑仍然表現出良好的C1-C2耦合能力以增強C3產物的形成,這歸因于重構的高密度Cu(111)/Cu(100)界面。

(a) FE for different products produced by HF-Cu-x under a range of pressure. (b) n-propanol partial current densities on different electrodes at various pressures and comparison of FEn?propanol/FEC2+ratios on different electrodes at various potentials. (c) FE for different products produced by HF-Cu-3 under a range of potentials. (d) FE for different products produced by HF Cu-3 under a range of KOH concentrations. (e)The Cu(111)/(100) interface per area, measured from the HRTEM images of HF-Cu-1, HF-Cu-3 and HF-Cu-10, plotted with the FEn?propanol/FEC2+ ratios. (f) FE for different products during CORR with HF-Cu-1 under 1 atm CO, HF-Cu-1 under 3 atm CO and HF-Cu-3 under 3 atm CO, respectively. The “1” in HF-Cu-1 denotes that the catalyst was electrochemically reduced and reconstructed under 1 atm of CO. (g) FEn?propanol during 130 h operation of CORR at a constant current density of ?100 mA cm?2
HF-Cu催化劑上的加壓CO電還原通過C1–C2耦合動力學和CO誘導的動態表面重構協同增強。CO分壓升高不僅會提高CO濃度,還會觸發動態結構重組,在3個atm的CO壓力下產生Cu(111)/(100)界面密度,是環境條件下的1.7倍。這種雙重機制使正丙醇在-0.67 V時的FE為28%。這些發現表明,CO壓力、界面位點密度和產物選擇性之間的相關性為了解C-C耦合動力學提供了見解。未來的研究應優先探索這種壓力調制重建策略在多碳產物合成中的普遍性,并優化氣體擴散電極結構,以減輕高壓下的析氫反應。