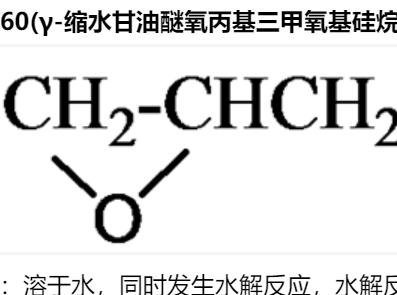
Barton等于 1986 年報道了一類制備芳胺類化合物的溫和方法[1],[2],即用芳基鉍試劑與脂肪胺或芳胺在銅或二價銅鹽的催化下常溫攪拌即可高產率的生成目標芳胺類 化合物。
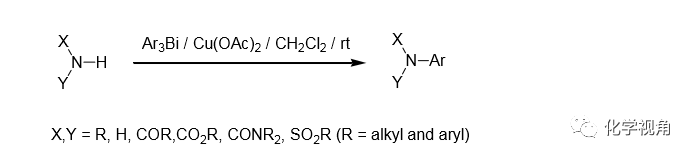
此反應對脂肪胺,芳胺有很好的收率, 對于一些非活性胺也有很好的收率,如 吲哚 ,酰胺,脲,咪唑,磺酰胺等。有報道稱用三乙胺或吡啶作堿可促進反應。此反應常用溶劑為二氯甲烷,常用催化劑為醋酸銅,常溫下反應。
芳基鉍試劑的制備通常是鹵代芳烴的格氏試劑和氯化鉍交換得到。
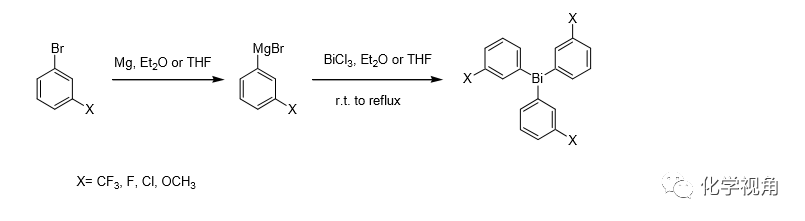
芳基鉍試劑的典型操作
Bromo-3-trifluoromethylbenzene (5.0 g, 0.0222 mol), dissolved in dry Et2O (100 mL) was added dropwise to a mixture of magnesium turnings (0.594 g, 0.0244 mol) and I2 (0.001 g) in Et2O in dry flask under N2 and a gentle reflux was maintained for 1 h. The reaction mixture was then cooled to 0 ℃and dry bismuth trichloride (3.50 g, 0.0111 mol) was added. After 10 min the mixture was warmed to reflux with stirring for 3 hrs. Water was added and the organic phase was separated, washed with water, dried (MgSO4) and concentrated under reduced pressure. The residue was chromatographed on a silica gel column (eluent: hexane/EtOAc 95/5) to give compound as yellow oil; yield, 52% 。
芳基鉍試劑芳胺化典型操作:
A slurry of amine, triarylbismuth (1-2 eq.), anhydrous Cu(OAc)2 (1-1.5 eq.), the tertiary amine (0.1-1.5 eq.) in methylene chloride (~2.5 mL/mmol of substrate) was stirred at room temperature for 6-48 hrs. The products were isolated by direct flash column chromatography of the crude reaction mixture on silica gel. The progress of reaction could be monitored by TLC as well as by the color change from the initial deep blue to turquoise green, with the concomitant precipitation of grayish Cu(I) salt. Additional amounts of Ar3Bi and Cu(OAc)2 could be added during the course of the reaction to ensure complete conversion of the substrate.