與金屬離子載流子相比,質子(H?)具有摩爾質量極低、離子半徑小、成本低廉及環境友好等優勢,是實現高功率密度和長壽命儲能的理想非金屬電荷載體。然而,開發兼具高容量、高穩定性和快速動力學的質子存儲正極材料仍面臨巨大挑戰。普魯士藍類似物(PBAs)雖具有開放的框架結構,但其本征電導率低,多金屬協同與結構穩定性難以兼顧。近年來,過渡金屬中的“d-p軌道雜化”與“d電子補償”策略在能源轉換領域展現出巨大潛力,但其在儲能材料設計中的應用尚未得到充分重視。開發通過原子級電子結構調控來協同提升PBAs材料導電性、活性與穩定性的策略,對于構建高性能質子存儲器件具有重要意義。 近日,華中科技大學盧興教授(通訊作者)、張盼盼教授(通訊作者)等人報道了一種原子級 d-p 軌道雜化策略,用于調節過渡金屬(V/Fe)的 d 軌道中心。通過原位共沉淀法合成了六氰合鐵酸釩(VHCF)/氧化釕量子點(RuOxQDs)異質結構(VHCF-RuOxQDs)。Ru 的 4d 軌道與 VHCF 的 C≡N 2p 軌道(氰基)的 d-p 雜化誘導了 π 反饋,并為調節 V/Fe 的 d 電子創造了“電子高速公路”,使它們的 d 軌道中心發生偏移,從而實現連續的多電子轉移。優化 d 電子結構降低了V5+ 的比例,從而減少了循環過程中的釩溶解。VHCF-RuOxQDs 正極在 1 A g?1 時具有 162 mAh g?1 的大容量,在 40 A g?1 時具有出色的倍率性能(127 mAh g?1),并且在超過 10000 次循環中具有超長的穩定性。當與 MoO3-MXene 陽極搭配時,這種非對稱全器件在 1.3 KW Kg?1的功率密度下實現了 53 Wh Kg?1的高能量密度。 VHCF-RuO?QDs通過原位共沉淀法合成,采用SEM、HR-TTEM、XRD、XPS、XAS等測試手段對其結構和形貌進行表征,結果表明RuO?QDs均勻分散在VHCF框架中, Ru與C≡N 之間的電子傳導,有效調控了V/Fe的d電子結構,形成了高效的“電子高速公路”。該異質結構具有優化的介孔結構和顯著提升的比表面積,并且錨定RuO?QDs不會破壞VHCF的原有晶體結構。(圖1)
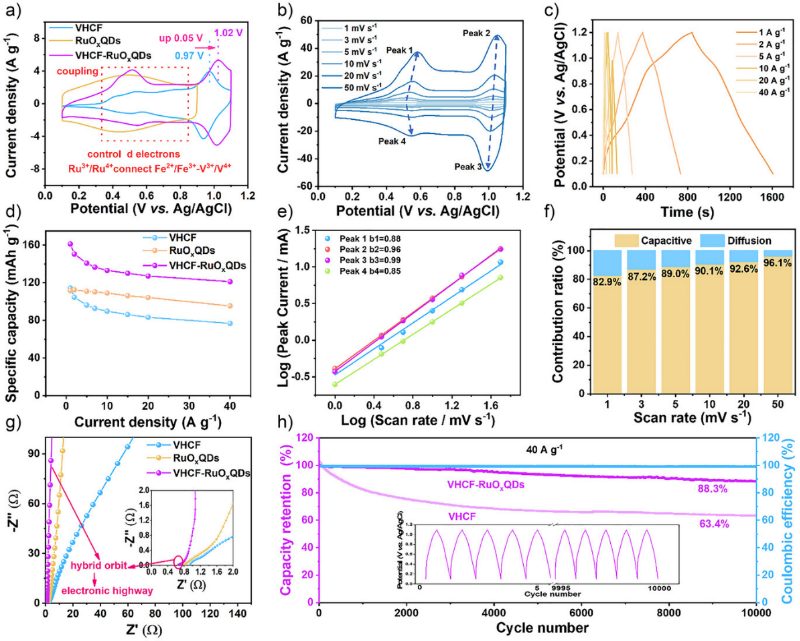
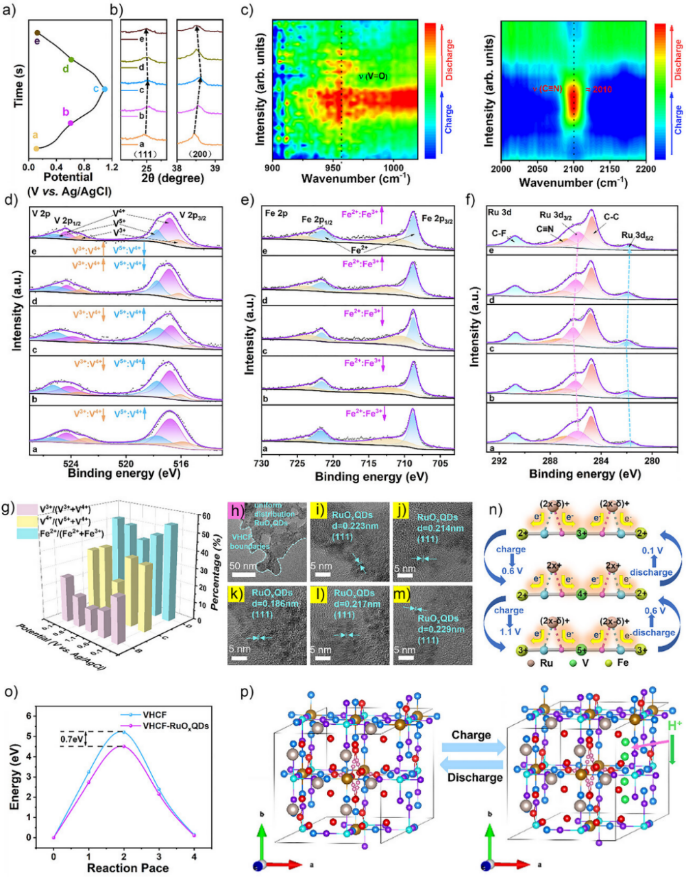
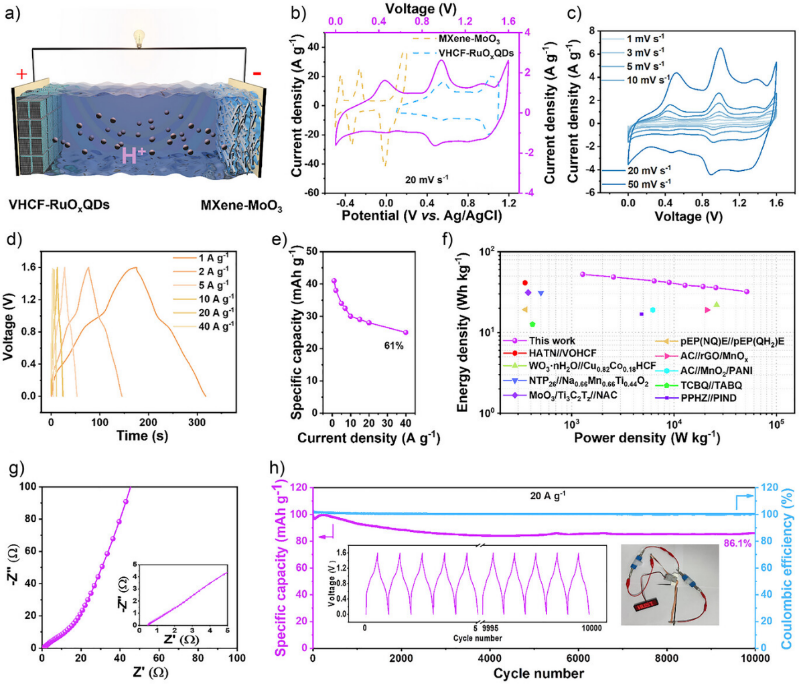
Figure 1. Synthesis and characterizations of VHCF–RuOxQDs. a) Schematic diagram of the “in situ co-precipitation synthesis” method for preparing VHCF–RuOxQDs and three-level magnified local structural diagram of VHCF and VHCF–RuOxQDs. b)–d) HR-TEM images of VHCF–RuOxQDs with different magnifications. e) and f) HAADF and EDS elemental mappings of VHCF–RuOxQDs. g) EXAFS and h) XANES spectra analyses of RuO2, Ru foil, RuOxQDs, and VHCF–RuOxQDs. i) FT-IR spectra of VHCF and VHCF–RuOxQDs. j) XPS valence band spectra of VHCF and VHCF–RuOxQDs. k) V 2p Fe 2p, and C 1s-Ru 3d XPS spectra of VHCF and VHCF–RuOxQDs. 構建了負載 RuOxQDs 的 VHCF 理論模型以及純 VHCF 模型以作對比,然后基于密度泛函理論(DFT)計算了它們的差分電荷和態密度。計算結果表明,VHCF 中 Ru 的 d 軌道與 C≡N 的 p 軌道之間發生了 d-p 軌道雜化,從而產生了 π 反饋效應和電子從 Ru 向 VHCF 的轉移,VHCF 的電子流入使 V/Fe 金屬中心充滿,激發了 C≡N-V 的 π 給電子效應,并抑制了 C≡N-Fe 的強電子排斥作用,進一步證明了通過外部原子級軌道雜化策略可以精確控制 PBAs 中過渡金屬的 d 電子結構。 Figure 2. Orbital hybridization structure of VHCF–RuOxQDs. a) Conformational optimization of VHCF–RuOxQDs. b) Differential charge density distribution of VHCF–RuOxQDs. The yellow region represents charge gain and the cyan region represents charge loss. c) HOMO and LUMO energy level electron variation of side view of VHCF–RuOxQDs. d) Schematic representation of d-band centers of RuOxQDs and VHCF–RuOxQDs. e) PDOS of C 2p, N 2p, and Ru 4d in VHCF–RuOxQDs. PDOS of f) Fe 3d, g) V 3d in VHCF and VHCF–RuOxQDs. h) d-p orbital hybridization mechanism for enhancing the conductivity–activity–stability of VHCF–RuOxQDs. 通過三電極系統對VHCF-RuOxQDs電極在3 M H2SO4電解液中進行電化學性能測試,循環伏安(CV)曲線顯示出多對清晰的氧化還原峰,對應于V3+/V4+、Fe2+/Fe3+和Ru(2x-δ)+/Ru2x+的連續多電子反應。通過動力學分析表明,其電荷存儲機制主要由表面贗電容行為主導(貢獻率高達96.1%)。該電極展現出優異的比容量(162 mAh g?1 @ 1 A g?1)和倍率性能(127 mAh g?1 @ 40 A g?1)。在40 A g?1的高電流密度下循環10000次后,容量保持率高達87.2%。(圖3) Figure 3. Electrochemical performance of VHCF–RuOxQDs electrodes. a) CV curves of VHCF, RuOxQDs, and VHCF–RuOxQDs electrodes at 5 mV s?1. b) CV curves at scan rates of 1–50 mV s?1 and c) GCD curves at different current densities from 1 to 40 A g?1 of the VHCF–RuOxQDs electrode. d) Specific capacities of VHCF, RuOxQDs, and VHCF–RuOxQDs electrodes calculated from the GCD curves as a function of current density. e) Log i versus log v plots for each redox peak in CV curves. f) Normalized contribution ratios of capacitive (orange) and diffusion-controlled (blue) currents as a function of scan rate. g) Nyquist plots of VHCF, RuOxQDs, and VHCF–RuOxQDs electrodes. The inset shows the magnified curves in the high-frequency range. h) Cycling stability and Coulombic efficiencies of VHCF and VHCF–RuOxQDs electrodes at 40 A g?1. The inset shows the first five and the last five GCD curves of the VHCF–RuOxQDs electrode. 為探明VHCF-RuO?QDs的儲能機理,我們對其進行了非原位XPS、XRD和原位FT-IR等測試,證實了V3+/V4+/V5+、Fe2+/Fe3+和Ru(2x-δ)+/Ru2x+作為氧化還原活性位點貢獻了連續的贗電容行為。非原位XRD表明質子嵌入/脫出過程為高度可逆的非相變反應,僅伴隨晶格的微小膨脹與收縮。結合DFT理論計算,Ru與C≡N的d-p軌道雜化形成了離域電子云,優化了電子傳輸通道,并將質子遷移能壘降低了0.7 eV,顯著提升了質子擴散動力學。(圖4) Figure 4. Proton charge–storage mechanism of VHCF–RuOxQDs electrodes. a) GCD curve of the VHCF–RuOxQDs electrode at 0.5 A g?1 and the corresponding b) ex situ XRD patterns, c) in situ FT-IR spectra, and ex situ d) Fe 2p, e) V 2p, and f) Ru 3d XPS spectra of the VHCF–RuOxQDs electrode at different charge/discharge potentials. g) Three-dimensional distribution of the valence occupancies of V and Fe at different charge/discharge states. h) TEM image of the VHCF–RuOxQDs electrode after 100 charge/discharge cycles. i)–m) HR-TEM images of the VHCF–RuOxQDs electrode at different charge/discharge potentials. n) Schematic diagram of the changes in valence states of Fe, V, and Ru during charge/discharge process. o) Migratory energy barriers of protons in VHCF and VHCF–RuOxQDs. p) Schematic illustration of proton charge storage in the VHCF–RuOxQDs electrode during charge/discharge process. 為證明VHCF-RuOxQDs潛在的商業應用價值,以其為正極、MoO3-MXene為負極、3 M H2SO4為電解液,組裝成非對稱全質子存儲器件。該器件擁有1.6 V的工作電壓窗口,實現了53 Wh kg-1的高能量密度(功率密度為1.3 kW kg?1)。在20 A g?1的高電流密度下循環10000次后,容量保持率高達86.1%,展現出優異的倍率性能和長循環穩定性,具有巨大的實際應用潛力。(圖5) Figure 5. Electrochemical behavior of MoO3–MXene//VHCF–RuOxQD asymmetric full device. a) Schematic illustration of the full device based on VHCF–RuOxQDs cathode and MoO3–MXene anode. b) CV curves of MoO3–MXene anode, VHCF–RuOxQDs cathode, and MoO3–MXene//VHCF–RuOxQDs at 20 mV s?1. c) CV curves of MoO3–MXene//VHCF–RuOxQDs at different scan rates of 1–50 mV s?1. d) GCD curves of MoO3–MXene//VHCF–RuOxQDs at different current densities of 1–40 A g?1. e) Calculated specific capacities at different current densities. f) Ragone plots of MoO3–MXene//VHCF–RuOxQDs in comparison with other reported devices. g) Nyquist plots and corresponding magnified curves in the inset of MoO3–MXene//VHCF–RuOxQDs. h) Cycling stability and Coulombic efficiencies of MoO3–MXene//VHCF–RuOxQDs at 20 A g?1. The insets show the first five and last five GCD curves and an energy-supply demo for powering an LED array of “HUST” logo. 論文信息 d-p Orbital Hybridization of Ternary Transition Metal Toward High-Performance Proton Storage Wei Tu, Ke Mao, Dr. Ying Huang, Jundong Shao, Xuan Tian, Pengfei Xu, Prof. Sheng Yang, Prof. Faxing Wang, Prof. Yao Gao, Prof. Panpan Zhang, Prof. Xing Lu Angewandte Chemie International Edition DOI: 10.1002/anie.202513523