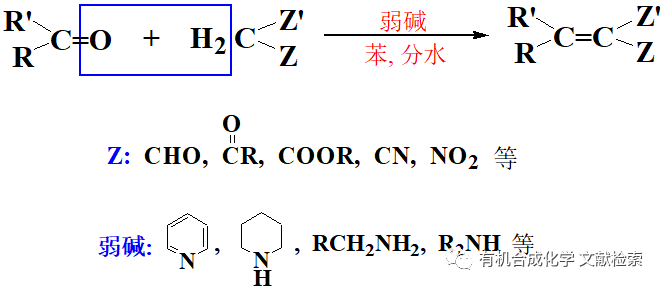
利用碳二亞胺類縮合劑縮合制備酰胺在藥物合成中應用極為廣泛,目前常用的縮合劑主要有三種:二環己基碳二亞胺(DCC)、二異丙基碳二亞胺(DIC)和1-(3-二甲胺基丙基)-3-乙基碳二亞胺(EDCI)。


常用的縮合活化劑有以下幾種,目前4-N,N-二甲基吡啶(DMAP)已被廣泛應用于催化各種酰化反應。有時在用DMAP催化效果不好時,可采用4-PPY,據相關文獻報道其催化能力要比DMAP高千倍左右。

目前,在藥物化學中用的最多的還是EDCI,其一個主要的特點就是其反應后的生成的脲是水溶性的,很容易被洗掉,一般EDCI與HOBt合用(注意: 這一反應HOBt一般是缺不了的,否則有可能導致縮合產率太低)。有時如果酸的a-位位阻大或者連有吸電子基團,反應會停留在活性酯這一步(這一活性酯的質譜信號較強,可通過MS或LC-MS檢測到)。由于HOBt也是水溶性的,其使得反應的處理和純化相對要容易。一般在這一縮合中要加入堿,特別當用胺或氨基酸的鹽酸鹽等縮合,常用的是加2-3當量的N-甲基嗎啡啉或二異丙基乙胺(DIEA,Hunig base), 縮合時以二氯甲烷為溶劑,若底物的溶解度不好,可用DMF作反應溶劑,再使用該方法進行。

1、DCC縮合法合成酰胺示例

To a solution of compound 11 (4.06 g, 10 mmol) in DMF (150 mL) wasadded N-hydroxybenzotriazole (HOBt, 5.64 g, 42 mmol), followed by dicyclohexylcarbodiimide (DCC; 8.60 g, 42 mmol). After stirring for l h,a solution of di-tert-butyl 4-amino-4-[2-(tert-butoxycarbonyl)ethyl]heptanedioate5 (17.34 g, 41.7 mmol)in DMF (60 mL) was added and the solution stirred at 25℃ for 23 h. The crystals were filtered and washed on thefilter with DMF (25 mL). The solvent wasdistilled at 50℃/1mm, and the residual oil was dissolved in ether (600mL). Crystals were filtered, the ethereal solutionwas washed successively with 10% HCl (2 x100 mL), saturated NaHCO3(2 x 100 mL), and brine (2 x 50 mL), then dried (Na2SO4). The ether solution was filtered throughcelite and solvent was then removed in vacuo to afford 19.0 g of crude product, which waspurified on a silica column eluting with toluene/EtOAc (1:1) to furnish (60%)the white, non-crystalline ester 12:14.0 g; mp 55-60℃. 1HNMR δ 1.43 (s, CH3, 108H), 1.94-2.28 (m, CH2CH2,64H), 5.87 (s, NH, 4H), 6.17 (s, CH=CH, 2H); 13C NMR (DMSO-d6) δ28.3 (CH3), 29.3 (CH2CH2),56.8 (CNH), 69.0 (CSO2), 171.1 (CO), 171.4 (CO).
2、DIC縮合法合成酰胺示例

To a solution of amine 13 (106 mg, 0.3 mmol),Fmoc-Phe-OH (116 mg, 0.3 mmol), and HOBt (44.8 mg, 0.33 mmol) in anhydrous DMF(2 mL) was added DIC (56 μL, 0.36 mmol). The resulting mixture was stirred at roomtemperature overnight, and DMF was then evaporated under high vacuum. The residue was dissolved in ethyl acetate (10mL), washed sequentially with saturated aqueous NaHCO3 and brine,and then dried over Na2SO4. The evaporation of the solvent gave the crudeproduct that was directly submitted for the Fmoc removal without purification. The crude product was dissolved in DMF (8 mL),piperidine (2.0 mL) was added, and the resulting solution was stirred at roomtemperature for 1 h. Following thesolvent evaporation, the residue was purified by silica gel chromatography (50%ethyl acetate in hexanes to 10% methanol in chloroform) to provide product 14(128 mg) in 85% yield as a mixture of two diastereomers. 1H NMR (CDCl3, 400MHz) ‰ 8.12-8.00 (2H, m), 7.90-7.80 (2H, m), 7.70-7.54 (2H, m), 7.42(2H, m), 7.35-7.20 (2H, m), 6.97-6.83 (3H, m), 5.32 (1H, m), 4.32 (1H, m),4.20-4.03 (2H, m), 3.96-3.80 (2H, m), 3.60 (1H, m), 2.98 (3H, m), 2.88 (2H, s),2.60 (1H, s), 2.00 (1H, m), 1.85 (2H, m), 1.77-1.55 (3H, m); MS (ES+) m/z) 501.4 (M + 1).
3、應用EDC縮合法合成酰胺示例一(二氯甲烷為溶劑)

To a solution of amine16 (0.284 mg, 1.19 mmol) and 5-hexenoic acid 15(0.136 g,1.19 mmol) in CH2Cl2 (12.0 ml) at 0℃were added HOBt (0.177 g,1.31 mmol) and EDC (0.251 g,1.31 mmol). The reaction mixture was stirredat room temperature for 10 h, then washed with 5% aqueous HCl (3×15.0 ml), 5% aqueous NaHCO3 (20 .0 ml),H2O (20.0 ml), and brine (20.0 ml), and dried (Na2SO4). Purification by flash chromatography(CH3Cl/MeOH, 10%, Rf = 0.43) afforded amidoalkene 17 in 99%yield as a brown oil.
4、應用EDC縮合法合成酰胺示例二(DMF為溶劑)

A DMF solution (10 mL) containing HOBt (103 mg, 0.76mmol), EDC (192 mg, 1.0 mmol), and Boc-D-Ile (172 mg, 0.76 mmol) was stirred atroom temperature for 20 h. A solution ofthe amino ketal 18 (0.41 g,0.76 mmol) and 4-methylmorpholine (0.17 mL, 1.5 mmol) dissolved in 10 mL of DMFwas then added to the reaction mixture. After4 h the reaction mixture was partitioned between EtOAc and H2O. The organic layer was washed with H2O,dried over MgSO4, and concentrated under reduced pressure. Flash chromatography (4:1 EtOAc/hexanes)afforded the Boc ketal 19 (0.44 g,0.59 mmol, 78%).
本文內容來源于網絡,版權歸原作者所有