有關碳水化合物的文獻中,烯丙醚用來保護醇是很常見的,原因在于烯丙醚通常可用各種方法形成糖苷。顯然,烯丙醚不能與強的的親電試劑共存,如溴、催化氫化的試劑。但它在中等強度的酸性條件(1N HCl,回流,10小時)下穩定。易于生成,在大量其它保護基存在下有許多溫和的脫保護方法,及其總體上的穩定性,使得烯丙醚成為許多反應系列中的主干。已有關于全氘代烯丙溴的合成及將它用作碳水化合物保護基方面的報道,全氘化合物的優點在于:烯丙基的NMR中的共振不再掩蔽其它可供鑒定的共振吸收峰,如糖苷的導頭碳的吸收峰。
烯丙基保護羥基示例(J.Org.Chem.C 1969,2367)
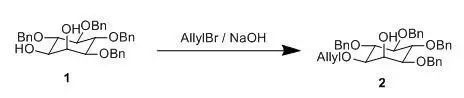
A mixture of 1 (50 g), powdered NaOH (50 g), benzene (500 mL), and allyl bromide (12 mL) washeated under reflux and the progress of the reaction was followed by TLC(ether: petroleum, 1:1). After 80 min. some starting material (Rf 0.15)remained and some diallyl derivatives present, but the major product was themonoallyl derivatives (Rf 0.56), the benzene solution was washed with water,dried (K2CO3) and evaporated. The syrupy product waschromatographed on alumina, elution with benzene:ether removed the diallylderivative (9 g) andelution with ether-methanol (25:1) gave the monoallyl derivatives (35 g ). Further elution with methanol gaveunchanged starting material (8 g).
烯丙基脫保護示例(J.Chem.Soc. PerkinsTrans 1, 1980,738)
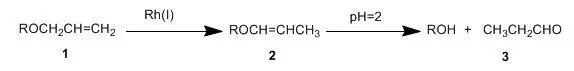
A solution of menthyl allyl ether (0.114 g, 0.58 mmol) (prepared from menthol,sodium hydride, and allyl bromide), RhCl(PPh3)3 (0.037 g, 0.040 mmol) anddiazabicyclo[2,2,2]octane (0.013 g,0.120 mmol) (added to inhibit premature hydrolysis of the intermediate enolether. Free propionaldehyde reacts with RhCl(PPh3)3 toform the catalytically much less active RhCl(PPh3)2CO.)in 10% aqueous ethanol was heated at reflux for 3 h. An aliquot was injectedinto 1 N HCl and after a few minutes was assayed by vpc analysis which showedonly menthol and menthyl allyl ether in 93% and 7% yield, respectively. Work upof a parallel reaction (by poured into water, extracting with ether, washingthe ether with brine acidified to pH 2, drying over MgSO4,concentrated, and separation on silica gel) gave menthol in 93% yield.
本文非原創內容,來源于網絡,版權歸原作者所有。