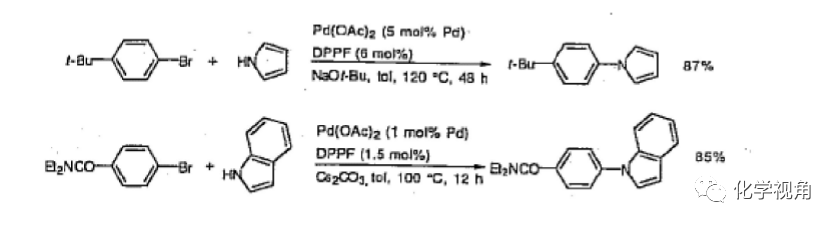
1、與吡咯及吲哚的反應
運用 dppf作配體,取代溴苯和吡咯及吲哚也能反應,而且有很好的收率。
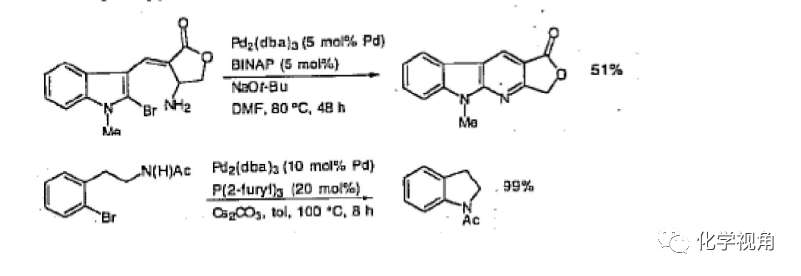
2、關環反應
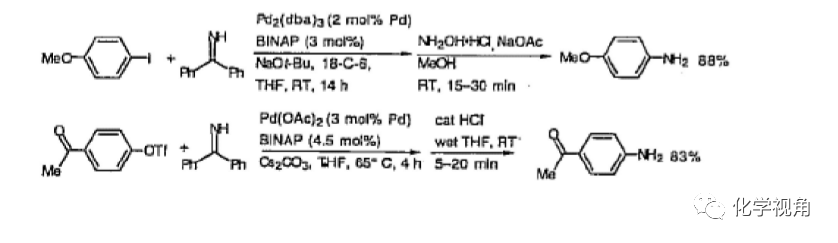
3、鹵代苯轉化為苯胺反應
二苯甲基亞胺與碘化苯或三氟甲磺酸酚酯在鈀催化下可高產率生成苯基亞胺,而二 苯甲基可在羥胺,醋酸鈉或鹽酸,四氫呋喃條件下溫和脫去。此方法為鹵代苯轉 化為苯胺提供個很好的途徑。
 4、Buchwald 反應操作示例
4、Buchwald 反應操作示例
Buchwald 反應常用的鈀催化劑為:Pd2(dba)3, Pd(OAc)2, 常用配體為:P(t-Bu)3, BINAP, P(o-tolyl)3, Xantphos, 常用堿有:Cs2CO3, t-BuOK,t-BuONa,常用溶劑有甲苯, 二甲苯,1,4-二氧六環。
催化劑和配體無固定搭配,常用效果較好的配體為 Xantphos 和 BINAP。對于底物
為苯環類化合物,溶解性較好化合物,常用甲苯作溶劑;對于雜環類反應,溶解性不好 的底物常用 1,4-二氧六環作溶劑。溴化物與胺的偶聯常用 t-BuOK 或 t-BuONa 作堿, 三氟甲磺酸酯與胺的反應常用 Cs2CO3作堿。
反應需在無水無氧條件下進行,一般回流反應。操作基本相似。
4.1 Buchwald 反應示例一
An oven-dried Schlenk tube was charged with cesium carbonate which had been finely ground with a mortar and pestle (1.4 eq) in a nitrogen-filled glovebox. The tube was capped with a rubber septum and removed from the glovebox. The tube was then charged with Pd2(dba)3 or Pd(OAc)2 and BINAP or PPF-OMe, and purged with argon. The aryl bromide (1.0 eq), the amine (1.2 eq), and toluene (2 mL/mmol halide) were added, and the mixture was heated to 100 oC with stirring until the starting material had been consumed as judged by GC analysis. The mixture was cooled to room temperature, diluted with ether (20 ml), filtered, and concentrated. The crude product was then purified by flash chromatography on silica gel.
4.2 Buchwald 反應示例二
Pd(OAc)2 (0.025 mol%) and P(t-Bu)3 or Xantphose (0.10 mol%) (Phosphine/Pd=4:1) were added to the suspension of aryl halide (40 mmol), diarylamine (40 mmol) and NaOtBu (48 mmol) in o-xylene (60 mL) in N2 atmosphere. The mixture was heated for 3 h at 120 oC. Subsequently, it was cooled to room temeperature. H2O (60 mL) was added to it, the organic layer was separated and concentrated. The crude product was then purified by flash chromatography on silica gel or re-crystallization with MeOH/THF.