酰鹵(酰氯、酰溴和酰氟)與氨或胺作用是合成酰胺的簡便的方法。通過酰氯、酰溴與脂肪族、芳香族胺均可迅速酰化,以較高的產率生成酰胺。但酰氟對水和其他親核試劑較為穩定。一般酰氯、酰溴與胺反應是放熱的,有時甚至極為激烈,因此通常在冰 冷卻下進行反應,亦可使用一定量的溶劑以減緩反應速度。常用溶劑為二氯乙烷、乙 醚、四氯化碳、甲苯等。由于反應中生成的鹵化氫,因此需要用堿除去鹵化氫,以防止其與胺成鹽。有機堿和無機堿均可用于此類反應,常用的有機堿有三乙胺、吡啶等, 常用的無機堿有 Na2CO3, NaHCO3, K2CO3, NaOH, KOH 等。在研究中我們發現,許多反應用無機堿反應更干凈且容易處理。
酰氯主要通過二氯亞砜和三氯氧磷的制備,高沸點的底物來說,二氯亞砜是合適的試劑;一般的酸在二氯亞砜回流數小時后,蒸掉過量二氯亞砜及溶劑后,再用些甲苯帶一下殘余的二氯亞砜即可用于下步反應。對低沸點的底物來說,則使用三氯氧磷較 為方便,主要由于低沸點很容易蒸餾出來。對于α-氨基酸,由于相應的酰氯在加熱會分解,因而一般不通過二氯亞砜和三氯氧磷的制備。當分子中有對酸敏感的官能團存在時,無法使用二氯亞砜,一般采用等當量的草酰氯和堿,一鍋發生酰氯再直接用于下步反應,近也有文獻報道應用三氯均三嗪在堿存在下可一鍋將酸轉化為酰氯。由于酰氯的活性太高,一般很難鑒定,有時為了判斷酰氯是否生成,一般取出一點加入過量的芐胺或甲 醇等,通過 TLC 確認反應進行的程度,也可以蒸干溶劑通過 HPLC, LC-MS 或 NMR 確 認。α-氨基酰氯一般也通過以上兩種方法合成。芳香酰氯相對烷基酰氯要穩定的多,例 如苯甲酰氯加入水中也要近半個小時才能分解完全。
酰氟要比酰氯穩定的,其對水和其他親核試劑都較為穩定,其可通過三氟均三嗪在吡啶的存在下制備(Tetrahedron lett. 1991,32(10),1303)。并可以通過層析分離出來。酰氟對水和其他親核試劑,易于保存和使用方便,我們有時可以利用它進行酰胺類化合物 庫的合成。
對于一些位阻較大活性很低的芳胺,往往即使用酰氯也有可能不發生反應,此時我們需要加入催化劑如 DMAP 等,有時也可不加任何堿直接由胺和酰氯高溫回流反應得到酰胺。
1、酰鹵的制備示例
1.1應用二氯亞砜合成酰氯實例。
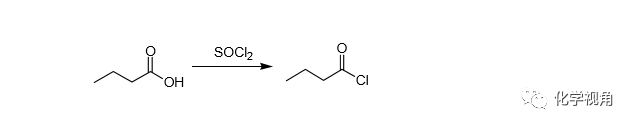
Fit a 100 mL two-necked flask with a dropping funnel and a reflux condenser connected at the top to a gas absorption trap. Place 36 g (21.5 mL, 0.3 mol) of redistilled thionyl chloride in the flask and 22 g (23mL, 0.25 mol) of butyric acid in the separatory funnel. Heat the flask gently on a water bath, and add the butyric acid during the course of 30-40 minutes. When all the acid has been introduced, heat was on a water bath for 30 minutes. Rearrange the apparatus and distillation: collect the crude acid chloride boiling between 70 and 110 ℃. Finally, restil from a flask provided with a short fractionating column and collect the butyryl chloride at 100-101 ℃. The yield is 23 g (86 %).
Note: Wrap a piece of absorbent cotton wool around the stem of the reflux condenser above the joint of the reaction flask to prevent condensed。
1、2 用草酰氯合成酰氯示例
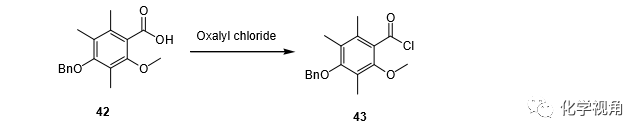
Compound 42 (4.19 g, 13.94 mmol) was dissolved in 20 mL CH2Cl , oxalyl chloride (4.25mL, 48.40 mmol) was added at room temperature, the resultant mixture was stirred at room temperature for 30 min and then the mixture was gently warmed under reflux for 30 min. After the mixture was concentrated in vacuo, the residue was dissolved in THF, and the solution was again concentrated in vacuo to offer quantitatively the crude chloride 43.
1、3 用三氯均三嗪合成酰氯示例
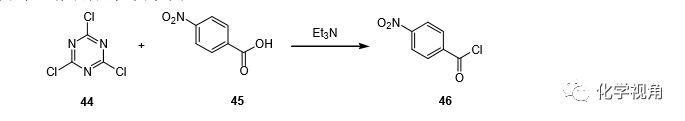
To a mixture of P-nitrobenzoic acid (1.67g, 0.01 mol) and 2,4,6-Trichloro-[1,3,5]triazine 44 (1.84g, 0.01mol) in acetone (20 mL) was added TEA (1.01, 0.01 mol) and the mixture was stirred for 3hr, filtrated to remove the separated dichlorohydroxy-s-triazine derivative, and the filtrate concentrated under reduced pressure. The residue was extracted with CCl4 and the solvent removed to give the desired acid chloride 46 (yield 58 %). (Note: the residue insoluble in CCl4 was found to be the unreacted carboxylic acid).
1、4 用三氟均三嗪合成酰氟示例
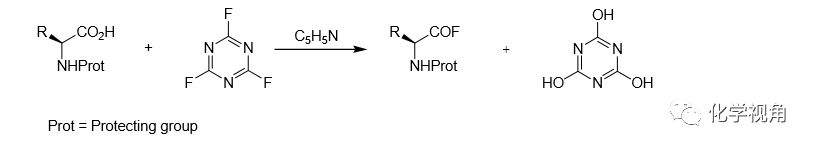
Typical experimental detail: equimolecular amount of the amino acid, pyridine and cyanuryl fluoride are mixed and stirred for 3~4 hours in dichloromethane at room temperature. At that time, ice-water is added to the reaction mixture and the precipitated canuric acid is filtered off the organic phase is dried and evaporated to dryness, which generally leaves the pure amino acid fluoride in crystalline form.