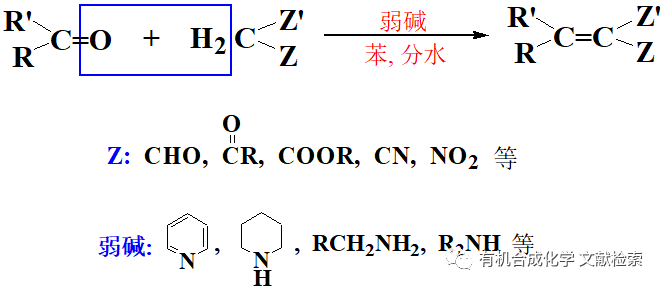
之醇氧化到醛酮
有機反應(yīng)中通常氧化反應(yīng)表現(xiàn)為分子中氧的增加或氫的減少。醇(包括伯,仲,叔醇)的氧化反應(yīng)是有機合成中經(jīng)常用到的反應(yīng)之一。醇用不同的氧化條件可以得到醛,酮或酸。下面就伯,仲醇氧化成醛酮做總結(jié);主要可以通過:鉻化合物,錳化合物,碘,釕化合物及DMSO氧化等。
氧化反應(yīng)之- 醇氧化成醛酮 | 鉻(VI) | 瓊斯試劑 (Jones Reagent): CrO3/H2SO4/H2O/Acetone | 易成酸 |
沙瑞特試劑 (Sarett Reagent) CrO3·2Py的吡啶溶液; 柯林斯試劑( CollinsReagents) CrO3·2Py的二氯甲烷溶液 | 易吸潮,不穩(wěn)定;需要無水條件反應(yīng) | ||
PCC(CrO3/HCl/Py) | 彌補collins 缺點 | ||
PDC(CrO3/Acetone/Py) | 中性條件反應(yīng) | ||
錳 | 高錳酸鉀(KMnO4) | 易成酸(雜) | |
二氧化錳(MnO2) | 選擇性好 | ||
活性DMSO | Pfizner-Moffatt氧化 | DCC/毒性大,不易除去 | |
Albright-Goldmannyang氧化 | (Ac)2O/代替DCC缺點 | ||
Parrikh-Doering氧化 | SO3-Py/反應(yīng)迅速 | ||
Swern氧化 | (COCl)2/低溫,副反應(yīng)少 | ||
碘 | IBX(DMP) | ||
釕(Ru) | TPAP | ||
Oppenauer/ TEMPO oxidation | |||
一:鉻的化合物
氧化醇的鉻化合物主要是六價鉻化合物,包括氧化鉻(CrO3,鉻酐),氧化鉻-吡啶配位化合物(Collins reagent), 氯鉻酸吡啶鎓鹽(PCC)等。這些試劑都需在酸性條件下反應(yīng),他們是使醇氧化成醛酮最普通方法。
CrO3是一種深紅色極易吸潮的固體,可以溶于水和一些有機溶劑,如叔丁醇,吡啶,醋酸酐等等,但在有機溶中不穩(wěn)定易爆。其在水中的存在狀態(tài)為。

醇的鉻酸氧化機理:醇和鉻酸作用首先形成鉻酸酯,隨后酯發(fā)生斷裂生成醛酮。

1.瓊斯試劑(Jones Reagent): CrO3/H2SO4/H2O/Acetone
瓊斯試劑氧化伯醇時一般不會停留在醛這一步,將繼續(xù)氧化為酸,機理如下:

Jones試劑,其制備方法是將26.72g CrO3 溶于23mL 濃硫酸中,加水稀釋到100mL。
瓊斯試劑氧化的一般操作步驟:
A 0.15–0.40 volume of H2SO4(c) is added over one volume of a 1.5–4.5 M solution of CrO3 in water. A fraction of the resulting red solution is dropped over a 0.01–0.5 M stirred solution of the alcohol in acetone. The alcohol causes the reduction of the red Cr (VI) cations to chromium species with a greenish look. A complete oxidation of the alcohol in a short time requires normally between 1.2 and 5.0 equiv. of CrO3. When the reaction is finished, the oxidant is quenched by the addition of 0.1–0.4volumes of 2-propanol. If so desired, the reaction mixture can be neutralized by the addition of saturated aqueous NaHCO3 or diluted NaOH. The resulting mixture is extracted with an organic solvent, such as EtOAc, DCM or Et2O. The collected organic solutions are washed with brine, dried (Na2SO4 or MgSO4).
2.柯林斯試劑(Collins Reagents:):CrO3·2Py
沙瑞特試劑為CrO3·2Py的吡啶溶液;
柯林斯試劑為CrO3·2Py的二氯甲烷溶液。制備這兩種試劑時,應(yīng)當(dāng)是將三氧化鉻加入到吡啶中,這個過程中伴隨著大量的放熱,反加可能會發(fā)生爆炸,得到的試劑也極其易爆,并有很強的吸水性,因此做反應(yīng)時一般要求惰性氣體保護。此類氧化中,用的溶劑一般是二氯甲烷,最好不要用沒有文獻報道的其他溶劑(可能會爆炸)。反應(yīng)中常常會加入一些硅膠,這樣可以減少鉻酸鹽沉淀對產(chǎn)品的吸附導(dǎo)致的損失。另外醇的β位含有雜原子時,容易發(fā)生β消除的副反應(yīng),加入乙酸酐可以有效阻止此副反應(yīng)的發(fā)生。
由于整個體系中是無水環(huán)境,因此此類試劑可以把伯醇氧化為醛時,不能形成縮醛,因此不會繼續(xù)氧化,可以停留在醛這一步。
但是Collins試劑存在較大的缺點:易吸潮,很不穩(wěn)定,需在無水條件下反應(yīng)。為使反應(yīng)完全及加快,需要過量的試劑。配置時容易著火。
氧化的一般操作步驟:
One equivalent of CrO3 is slowly added over a 0.2–2.0 M solution of 2–2.03 equivalents of dry pyridine in dry DCM. Very often, ca. 2–7 g of dry Celite per g of CrO3 are added-normally before the preparation of the CrO3·2Py complex-in order to avoid loss of product on the chromium precipitates during the work-up.
Very frequently, ca. 2–5 equivalents of acetic anhydride are added-normally after the preparation of the CrO3·2Py complex-in order to facilitate a milder reaction, particularly in sugars and nucleosides. It is not common to add both Celite and acetic anhydride in the same reaction.
After ca. 15–20 min, a 0.02–0.70 M solution of the alcohol in dry DCM is slowly added. Normally, between 4 and 10 equivalents of the CrO3·2Pycomplex are used per equivalent of alcohol. When most of the starting alcoholis consumed, two alternative work-ups can be carried out.
Work-up A:
The reaction mixture is filtered through a pad of silica, Florisil or Celite. The filtrate is washed with an organic solvent, like Et2O, EtOAc, or DCM. The collected organic phases may be optionally washed with diluted HCl, diluted aqueous base, and brine or saturated CuSO4 solution. The resulting organic solution is dried (Na2SO4 or MgSO4) and concentrated.
Work-up B:
The reaction mixture is sequentially washed with NaOH (5%), HCl (5%), NaHCO3 (5%) and brine. Adding some ether can help the fractioning. Optionally, the organic phase can be subsequently filtered through Florisil. The result in gorganic solution is dried (Na2SO4 or MgSO4) and concentrated.
3.PCC(pyridinium chlorochromate)
PCC法基本彌補了Collins法的所有缺點;成為目前最為廣泛的醇氧化成醛酮的方法。

制備方法:將三氧化鉻(1eq)加入到6M鹽酸(1.1eq)中得到氯鉻酸溶液,0℃下分批加入吡啶(1eq)后過濾得到橙黃色的PCC固體,易于儲存和操作。
氧化的一般操作步驟:
Approximately, 1.1–7 equiv., typically1.5 equiv. of solid PCC is added over a ca. 0.01–0.25 M solution of the starting alcohol in dry DCM. The resulting mixture is stirred at room temperature. Very often, ca. 0.2–1.2 g of activated MS per mmol of alcohol are added in order to accelerate the reaction. In order to moderate the acidity of PCC, it is very common to add ca. 0.3–1 equivalents of NaOAc. A solid support, such as silica gel, Celite, Florisil or magnesium sulfate, is added, very often in a proportion of ca. 0.3–2 g of solid support per mmol of alcohol, in order to facilitate the work-up. Occasionally alumina, working both as a solid support-used to facilitate the work-up and as an accelerant, mixed with PCC is added, in a proportion of ca. 0.4–1.5 g of alumina per mmol of alcohol. Normally, PCC is deposited over the alumina. Occasionally, ca. 10–20 equivalents of acetic acid are added in order to accelerate the reaction.
Sometimes, the reaction flask is sonicated with ultrasound in order to fragment the surface of the PCC particle sand, therefore, accelerate the reaction.
Although in PCC oxidations, it is very common to add simultaneously to the reaction an accelerant, a buffer and a work-up-facilitator; it is not common to employ simultaneously two materials belonging to the same kind, with the exception of the combination of the two accelerants molecular sieve and acetic acid, which are very often used together.
Work-up:
When a TLC analysis shows that most of the starting alcohol is consumed, the solids suspended in the reaction and the chromium species are removed by filtration through a pad of Florisil1,silica gel, alumina or Celite, and the pad is washed with an organic solvent, such as ether, DCM, or EtOAc. Sometimes, the solids can be removed by decantation. Other times, it is advisable to add some diethyl ether to the reaction mixture before the filtration, in order to promote the separation of reduced chromium species in a granular form. Occasionally, the reaction mixture is c